Zeno Experiment Overview

Zeno Experiment Overview
Thermodynamic critical points are ubiquitous, but not easy to observe. Although critical opalescence was first seen in 1869, it is only within the last 25 years that the study of critical phenomena has become a recognizably separate field of physics. During that time its growth has been rapid, with much success understanding the nature of the critical point.
Critical points command attention because they are very simple, nearly ideal systems which nevertheless exhibit a richness of behavior. Associated with every critical point is a quantity called the order parameter, which is an intensive thermodynamic property of the system. The order parameter has the simple property that, along some thermodynamic path which passes through the critical point, its value is zero on one side of the critical point, while on the other side it is nonzero but vanishes smoothly as the critical point is approached. If you classify critical points according to the type of their order parameter and the spatial dimension of the system, the Universality Hypothesis asserts that each of these universality classes will have the same critical point behavior.
Near a critical point, a system's thermodynamic properties are strikingly anomalous; for example, the heat capacity and the isothermal compressibility in fluids become infinite. With distance from the critical point measured in terms of appropriately scaled parameters, such as temperature or pressure, these divergent quantities can be successfully described with power-law functions: the distance parameter is raised to some power, and each quantity has a characteristic exponent. The Universality Hypothesis predicts that these power-law exponents for different systems belonging to the same universality class should have identical values. Thus, the Universality Hypothesis focuses attention on critical exponents as the primary means of describing critical points. In a system far from any critical point, its macroscopic character is an expression of intermolecular interactions; the crystalline structure of a material is determined by microscopic forces and molecular structure, as are the properties of the fluid. However, near a critical point, molecular details lose importance as the physical nature of the system is dominated by thermodynamic (equilibrium) fluctuations of the system's order parameter. The closer the system is to its critical point, the larger the fluctuations become, both in magnitude and in spatial extent. Fluctuations, which are normally so small that they have a negligible effect on the system, grow to such an extent that they impinge on the macroscopic world. Encompassing enormous numbers of molecules, these fluctuations hide the details of intermolecular forces and the system takes its characteristics from the fluctuations instead. It is the simplicity of these large fluctuations which allows a wide variety of physical systems to exhibit identical critical point behavior.
The study of critical phenomena is the study of the fluctuations. Describing the static and dynamic properties of the fluctuations should lead directly to understanding the thermodynamic properties of the critical system. Furthermore, the Universality Hypothesis says that any members of a universality class should be representative of all the others, although this remains a conjecture which must be tested experimentally. We have designed an experiment which will measure two fundamental dynamic properties of critical fluctuations: their spatial extent (or correlation length) and their lifetimes (or decay times). The system we have chosen to study is the simple fluid xenon near its liquid-vapor critical point, and the experimental technique is light-scattering spectroscopy. The technique and the system are well-suited to each other: light-scattering measurements can be made with very high precision, and light is a nonintrusive probe which couples directly to the density fluctuations of the fluid system. Light-scattering studies in fluid systems have provided our best measurements of dynamic critical point behavior, but experimental progress in pushing closer to the critical point in simple fluids has all but stopped because of the limitations imposed by gravity.
The problem with gravity results from the same thing that makes critical points so interesting, namely the divergence of thermodynamic properties. In particular, it is the diverging compressibility that becomes a problem in a fluid system. The power-law behavior means that, in order to separate critical properties from normal properties, the system must be very close to its critical point, in the ``asymptotic'' region, so that the critical point response is clearly measured. In a typical fluid system, the asymptotic response begins to emerge at temperatures about 1 mK to 100 microKelvin away from the critical point. However, as the asymptotic region is approached, the compressibility of the fluid becomes so large that the weight of the fluid itself causes severe compression of the fluid. So great is the compression that only a thin layer of fluid remains which still shows critical-point behavior. The closer the system is to the critical point, the thinner this layer of critical fluid becomes. Even light scattering, which is capable of sampling a very small region of the fluid, becomes ineffective just when the asymptotic region is being reached.
The very low gravity environment of low-Earth orbit will alleviate the compressibility effects, allowing these measurements to approach 100 times closer to the critical point. We note that the correlation length and decay times are fundamental properties of the critical fluctuations, not tied to any particular model or theory of critical phenomena for their interpretation. Thus, careful, high-precision measurements of these properties in a low-gravity environment will provide a data set with lasting value to further our understanding of critical phenomena.
The Zeno Instrument
The Zeno instrument is contained in two parts: the Optics
Module and the Electronics Module. Both are mounted on
the USMP carrier in the cargo by of the Shuttle. Together,
the modules weigh about 1000 pounds.
The Electronics Module contains most of the electronics
for the experiment, except for some temperature-sensitive
elements which are in the Optics Module. Included is
a complete computer system (a single-board computer with
a 68020 processor running an OS-9 operating system), and
a specially-designed digital correlator.
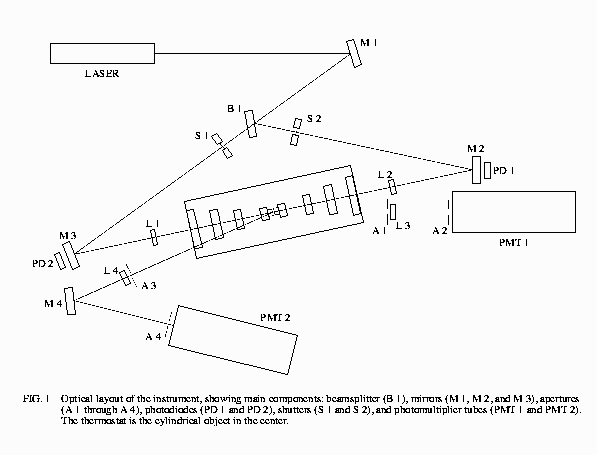
The Optics Module contains the light-scattering spectrometer
on an isolated optics bench. The optical layout of the
instrument is shown in the figure below.
Central to the experiment is the multi-shell thermostat
which contains the xenon sample cell; this thermostat, in
the controlled environment of the Optics Module, is able
to control the sample temperature with a precision of
3 microKelvin. The optical windows of the sample cell
are arranged such that the optical path length through
the xenon is only 100 microns; in other words, we scatter
light from a very thin sample of fluid.
In a nutshell, light from the laser enters the sample
from one of two directions, determined by the shutters.
(The two paths contain different filters in the beam,
so that selecting different paths gives us a factor
of 10 change in incident laser power.) The fluctuations
in the fluid scatter light, some of which is collected
at specific angles by the two PMTs, and processed by
the digital correlator to give us information about
the decay times of the density fluctuations.
In addition, photodiodes behind the partially-reflecting
mirrors before and after the thermostat are used to
measure the extinction of the beam as it passes through
the sample, due to the increase in the turbidity of
the sample as it nears the critical temperature.
Measuring the turbidity of the sample gives us information
about the size (correlation range) of the fluctuations.
The Experiment
The experiment itself is simple, but proceeds carefully
and slowly.
Starting at a temperature well above the critical temperature (Tc),
we change the temperature of the sample by small increments,
moving between temperatures with slow temperature ramps. The
starting point is about Tc + 4K, and then we move towards
Tc in geometric progression: Tc + 1K, +.6K, +.3K, +.2K, +.1K,
+.06K, ..., until we get to Tc + 100 microKelvin.
At each temperature set point, we pause for several hours,
wait for the system to reach stability, and take a series
of turbidity and decay-rate measurements, and then move on
to the next point.
As we move through these temperatures, we model the data
we're getting to give us ever-better estimates of the
critical temperature, better estimates which we use to
get closer, to get better estimates, to get closer,...,
until at the end of the experiment we will cross through
the critical point and collect data to give us a
high-precision location on our system thermometry.